Isotope Production and Application
Professors
- SATO, Tetsuya, e-mail: sato.tetsuya.6d<atmark>kyoto-u.ac.jp
* replace <atmark> with @
For chemists, the periodic table is akin to “a map”. How far does it go? What are the chemical properties of the heaviest elements at the very end of the table? The seventh period is now complete, and we are moving toward the eighth, making these questions even more exciting. In fact, it is suggested that the heaviest elements may not follow the standard periodic laws.
We can also look at the periodic table in a different way by focusing on neutron numbers. This reveals isotopes – atoms with the same atomic number but different mass numbers. When isotopes are far from stability, their nuclei become unstable and release radiation as they decay. This radiation carries far more energy than a typical chemical reaction. This powerful energy is useful not only for highly sensitive analysis but can also act as a “scalpel” to target and destroy cells with great precision.
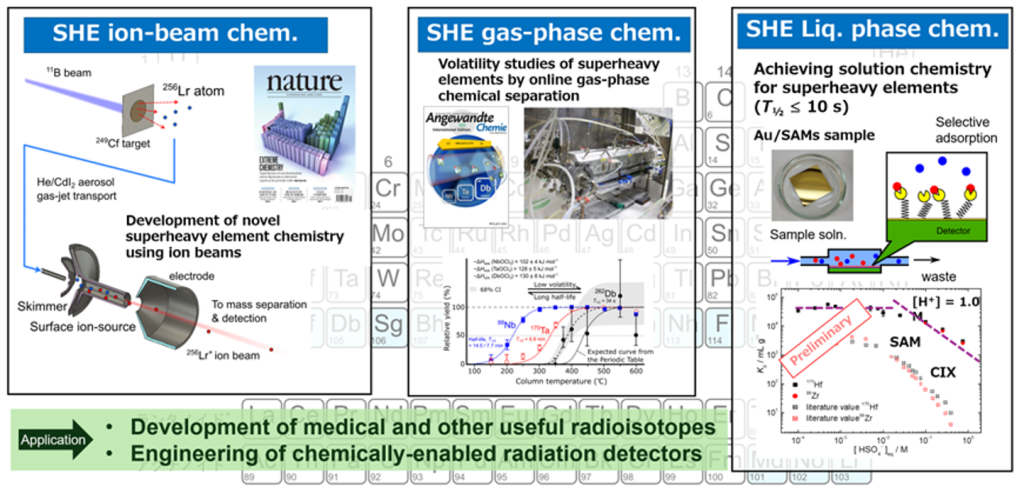
In our laboratory, we conduct ion beams, gas-phase chemistry, and solution chemistry to understand the properties of superheavy elements. We also utilize nuclear reactions to synthesize radioactive isotopes and perform chemical separation of them. As an application, we are developing useful radioisotopes, including for medical use, and engineering chemically-enabled radiation detectors.